| Take
action to stop these violations of the International
Code of Marketing of Breast-milk Substitutes.
The people responsible have names and addresses - call on them to
market their products ethically.
The tables below give
details of some recent violations. The date when the violation
was last reported to Baby Milk Action or confirmed to be current
is given. The violation reference is for Baby Milk Action's records.
Please quote it if forwarding correspondence to us, if possible.
Read
company responses received so far
Nestlé
launches infant formula promotion campaign in southern Africa,
violating Resolution on HIV
|
Company
|
Item
|
Date
|
Violation
Reference
|
|
Nestlé
|
Promotion of infant
formula
|
On-going
|
comp/01/11
|
Background:
|
Nestlé
is using the suffering of infants infected with HIV as
a cover to launch promotion of infant formula in southern
Africa. Research has shown that exclusively breastfed
infants are at no more risk of infection than artificially-fed
infants. Increased risk arises when breastfeeding mothers
introduce formula or other substances (see Update
29). This is more likely to happen following the irresponsible
promotion launched by Nestlé.
For many years
Baby Milk Action has been calling for independent research
into the risks of mother-to-child-transmission of HIV
through breastfeeding. The importance of this was acknowledged
at the World Health Assembly this year in Resolution
54.2.
|
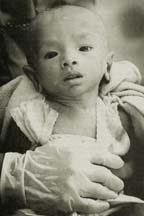
6-month-old
Roath Chamrouen abandoned at birth and HIV-positive,
cared for at Phnom Penh Nutrition Centre, Cambodia,
Photo taken from the Bangkok Post, August 11, 1999.
See IBFAN's briefing paper on The
International Code, HIV and Breastfeeding.
|
Resolution 54.2 calls
on Member States:
"to recognize
and assess the available scientific evidence on the balance
of risk of HIV transmission through breastfeeding compared
with the risk of not breastfeeding, and the need for independent
research in this connection; ... and that those who choose
[options other than breastfeeding] should be encouraged to
use them free from commercial influences;"
Governments barely
had time to act on the Resolution when Nestlé announced
a new infant formula promotion campaign in southern Africa which
violates many aspects of the Resolution (see Business
Report, South Africa, 3rd August 2001). Nestlé is
launching a "Nutrition Institute". According to Nestlé's
Ferdinand Haschke, "The long term goal is to improve nutrition
in southern and east Africa, in particular nutrition used in
the HIV vertical transmission programme through infant formula."
Nestlé is promoting
its Nan Pelargon formula in southern Africa and claiming
that its high acidity means it will destroy infections in unsafe
water. Independent health experts asked by Baby Milk Action
suggest that such a claim cannot be supported.
Nestlé's initiative
undermines the requirement that mothers receive independent
advice 'free from commercial influence.' Nestlé's press statements
suggesting its formula can be used with unsafe water is a thinly-disguised
promotion strategy and puts all infants at risk.
Nestlé ignores
research evidence from South Africa which has shown that exclusively
breastfed infants of HIV-infected mothers are at no more risk
of infection than exclusively artificially-fed infants. Increased
risk occurs when breastfeeding mothers introduce other substances.
UNICEF has estimated that in the last 20 years 1.7 million babies
may have contracted HIV from their mother's breastmilk, while,
in the same time, 30 million babies have died because they were
not breastfed. Breastfed infants are at much lesser risk of
diarrhoearial disease, respiratory infections and other illnesses
and their mothers also experience health advantages.
Suggested letter
to the man responsible: Peter
Brabeck-Letmathé, CEO, Nestlé S.A., Av. Nestlé
55, CH-1800 Vevey, Switzerland. Fax: + 41 21 924 2813.
|
As you know,
the World Health Assembly adopted Resolution 54.2 in May
2001 stressing the "need for independent research"
into "the balance of risk of HIV transmission through
breastfeeding compared with the risk of not breastfeeding."
The Resolution
also states that mothers who decide not to breastfed and
choose other options "should be encouraged to use them
free from commercial influences."
It is very
disappointing that Nestlé almost immediately decided to
violate the Resolution by launching a "Nutrition Institute"
in southern Africa to promote Nan Pelargon formula
to health workers for HIV interventions. By definition,
Nestlé's information is not independent.
It is also
very disturbing that Nestlé staff are promoting Nan
Pelargon infant formula in the media as suitable for
use where water supplies are unsafe.
Will Nestlé
take full responsibility for all deaths that occur through
misuse of this formula?
I request you
abide by Resolution 54.2 and stop these promotion campaigns
immediately.
|
Wyeth's
aggressive promotion in South Africa
|
Company
|
Item
|
Date
|
Violation
Reference
|
|
Wyeth
|
Widespread and
systematic violations
|
Ongoing
|
comp/01/12
|
Background:
Wyeth's unethical
promotion - (left) SMA Gold "now even closer to breastmilk",
idealizing text because as an inert substance it is still far
away. (Right) In store promotion for the re-labelled SMA products
in South Africa.
Wyeth formulas have
been marketed in southern Africa under licence by Infacare.
Wyeth has always denied any responsibility for labelling and
other violations. However, it is now marketing its brands directly
and has used this change to launch an aggressive promotion campaign,
which has provoked a number of complaints. Nestlé, which has
launched its own infant formula promotion campaign in southern
Africa recently (see above), also reported Wyeth to the authorities.
This demonstrates the fierce and dirty war taking place between
baby food companies - in the US Wyeth observed a voluntary ban
on advertising until Nestlé took it and other companies to court
and began advertising itself!
 |
Magazine advertisements
in South Africa for Wyeth's S-26 brands (see left) which
undermined breastfeeding and idealized artificial infant
feeding were censured by the South African Advertising
Standards Authority in May 2001. In a written submission
the Department of Health stated the:
"picture
and text 'our babies are about to have a face lift...
implicates that babies will 'have a face-lift' with
the breastmilk substitutes and therefore negates the
superiority of breastmilk... In this regard, the Department
of Health, which is the principal custodian on child
survival issues, urges Wyeth to desist from contravening
the Code."
|
A television advertisement
showed a baby playing with a computer followed by a voice-over:
"Because I have changed my infant formula to S-26 Promil
Gold this computer is child's play". This implies that
babies fed on S-26 formula will be more intelligent.
Wyeth wrote to the ASA disputing that S-26 Promil Gold
comes within the scope of the South African Code of Ethics as
it is promoted for use from 6 months. This ignores the fact
that it will still replace that part of a child's diet best
provided by breastmilk - breastfeeding is recommended into the
second year of life and beyond by the World Health Organisation.
The S-26 brand name is used for Wyeth's infant formula
as well as follow-on formula and advertising one product serves
to advertise them all.
Suggested letter
to the man responsible: Mr. John R Stafford, CEO, Wyeth, PO Box
8616, Philadelphia, Pennsylvania 19101, USA. Fax: +1 610 688 6228.
|
Wyeth has
recently been censured by the South African Advertising
Standards Authority for its promotion of S-26 products
following a complaint by Nestlé and others. It is disappointing
that Wyeth has opposed the ruling. Can you confirm that
Wyeth will now accept the ruling and stop its unethical
and irresponsible promotion of these products?
Wyeth attempted
to argue that S-26 Promil Gold does not come
within the scope of the South African Code of Ethics
and similarly misrepresents the International Code
of Marketing of Breastmilk Substitutes and subsequent,
relevant World Health Assembly Resolutions.
The recent
IBFAN monitoring report, Breaking the Rules, Stretching
the Rules 2001, demonstrates that Wyeth violates
these measures in many other respects.
This attitude
may soon result in Wyeth and its parent, American Home
Products, being targetted by an action similar to the
popular Nestlé boycott, which is now active in 20 countries.
Boycott action
will be less likely if Wyeth indicates that it will
accept the baby food marketing policy of the World Health
Organisation (as set by the World Health Assembly Resolutions).
Will you
please provide a clear statement accepting that the
International Code of Marketing of Breastmilk Substitutes
and subsequent, relevant World Health Assembly Resolutions
are minimum requirements for all countries?
|
You
can be a Code Monitor.
|



